Example of electron capture8/14/2023 The creation of a positron requires 511 keV, the mass energy of the positron. 
However, electron capture is more economical in energy than positron emission, its competitor. The probability that an electron, even one belonging to the inner ‘K’ shell, would find itself inside the nucleus is very low indeed (for potassium 40, the volume of the nucleus is less than a billionth of the K layer volume). Whereas beta decay can occur spontaneously when energetically allowed, for an electron capture the weak forces require that the electron comes into close contact with a proton of the nucleus. Electron capture occurs much less frequently than the emission of a positron. Weak forces are behind positron emission and electron capture. 
This explains why electron capture is difficult and therefore rare.Įlectron capture : a difficult and rare process Even the innermost K-layer electrons are far from the very small volume of the nucleus where the weak forces responsible for the capture operate and transform the electron into a neutrino. Most of the electrons orbit the nucleus at distances large compared to the nucleus. The captured electron belongs to the group of electrons orbiting around the nucleus. Ordinary beta-minus decay has no competitor on Earth however to reduce an excess of neutrons, since the capture of positrons would occur in an world made of antimatter. Electron capture, along with beta-positive decay, is Nature’s way of guaranteeing that no nucleus becomes too proton-heavy. The capture of an electron has the same effect on a nucleus as the emission of a positron: one of its protons transforms into a neutron, diminishing the global electric charge of the nucleus by 1 unit. The electron capture trigger the emission of an invisible neutrino by the nucleus. The best-known example is of potassium 40 : 11% of the nuclei of that isotope of potassium present in our body decay by electronic capture. In both cases, practically all the enegy released is carried by light particles.Įlectron capture is a comparatively minor decay mode caused by the weak force. There is no such energy threshold in the case of electron capture (bottom). The emission of a positron and the capture of an electron are twin reactions both resulting in the diminution of the number of protons by 1 (from Z to Z-1) and the production of a neutrino.The positron observed in the final stage of the beta decay (top) is a new particle requiring the 0.511 MeV of its rest mass energy to be created. If this requirement is not met, then orbital electron capture takes place exclusively.Positron emission versus electron capture The other item is that the daughter product has one less orbital electron than the parent. One item is the positron ejected from the nucleus of the parent. This mass difference between the parent and daughter is necessary to account for two items present in the parent but not in the daughter. For positron emission to occur, however, the mass of the daughter product must be less than the mass of the parent by an amount equal to at least twice the mass of an electron. Electron capture and positron emission result in the production of the same daughter product, and they exist as competing processes. Electron capture is shown graphically in Figure 8. Also, there will always be characteristic x-rays given off when an electron from one of the higher energy shells moves in to fill the vacancy in the K-shell. 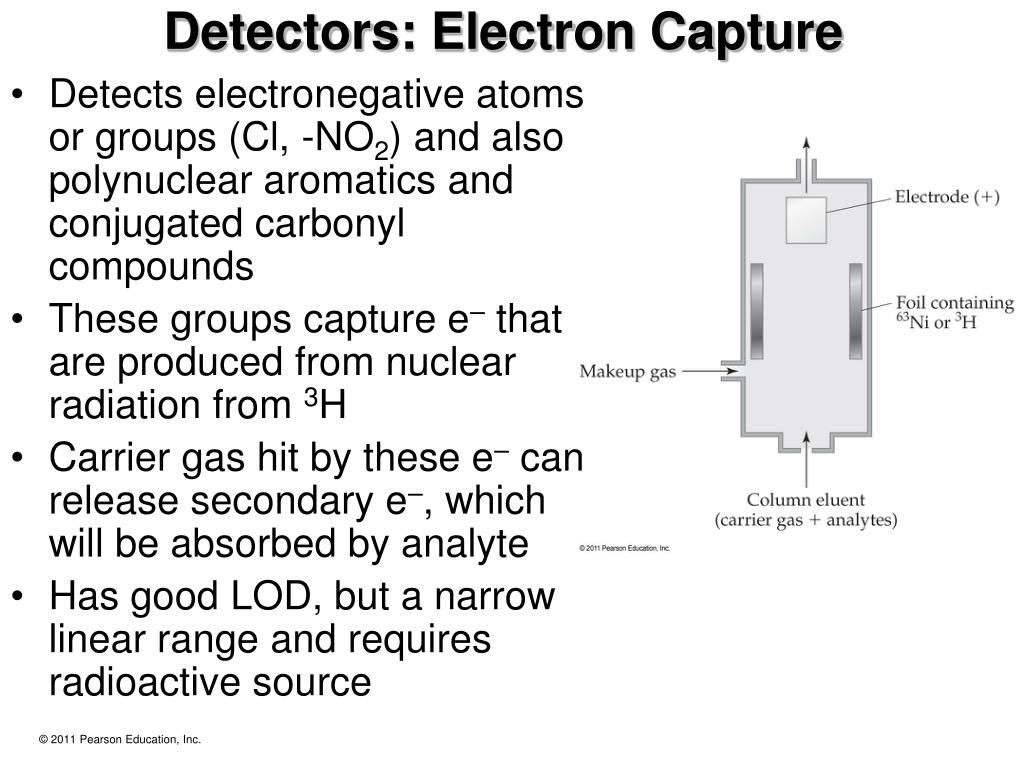 
Any energy that is available due to the atomic mass of the product being appreciably less than that of the parent will appear as gamma radiation. A neutrino is formed at the same time that the neutron is formed, and energy carried off by it serves to conserve momentum. The following example depicts electron capture. The electron is normally captured from the innermost orbit (the K-shell), and, consequently, this process is sometimes called K-capture. This process is called electron capture (EC). Electron Capture (EC, K - capture) Nuclei having an excess of protons may capture an electron from one of the inner orbits which immediately combines with a proton in the nucleus to form a neutron. An example of a typical positron (beta-plus) decay is shown below. A proton has been converted to a neutron. When a positron, represented as e,, or simply as +1 +1 0 0 e or, is ejected from the nucleus, the atomic number is decreased by one and the mass number + + remains unchanged. Except for sign, they are nearly identical to their negatively charged cousins. 0 NP-01 Figure 9 Orbital Electron Capture Positively charged electrons (beta-plus) are known as positrons. Atomic and Nuclear Physics DOE-HDBK-1019/1-93 MODES OF RADIOACTIVE DECAY Rev.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
